Ecology

Figure 1: Sampling site for focus experiment, approximately 1km offshore of Heron Island, Great Barrier Reef. (23’’27’538S, 151'' 556'47E.)
Focus Experiment
Introduction
Tropical holoplanktonic gastropods have been overlooked as important invertebrates within the Great Barrier Reef ecosystem,despite being the base of many marine food webs and dominating the GBR pelagic ecosystem (McKinnon et al. 2007). One of the reasons for the paucity in knowledge is their extremely delicate shells,which are of utmost importance for taxonomic identification (Newman 1990).
Shells are often damaged and pteropods are killed in long plankton tows. This study used a short, stationary, vertical tow to obtain as many specimens as possible, without damaging the shells. In addition, this study hoped to collect tiny Creseis veligers and so a fine mesh (100µm)was used. The aims of this study were to understand which parts of the tidal cycle, particularly on ebbing and flooding high tide contained the highest densities of the genus Creseis.
C. acicula is a tropical, epipelagic species that prefers neritic waters, though their interaction with tidal cycles is currently unknown. As they are primarily coastal organisms, it is expected that C. acicula will be affected by tidal currents, and may have ecological interactions and adaptations associated with this. This study aimed to observe the relative densities of C. acicula, and qualitatively observe the contiguous tidal planktonic community.
In addition, this study aimed to discriminate between the relative densities of adult and veliger forms of C. acicula to establish whether different life stages occupy different temporal niches within the water column. The adult and veliger forms differ in their physical characteristics,including different locomotion and feeding strategies. Creseis veligers feed and move using a ciliary mechanism and adults ‘flap’ their wings and capture prey using a mucus web. This study aimed to see if there were any differences in adult and veliger distribution throughout the tidal cycle, to establish whether the physical differences may also implicate ecological differences.
Understanding how currents and tides affect the zooplankton community may have implications for modelling the potential impacts of climate change,through indirect effects. These indirect effects include changes to the oceanographic processes that alter the mixing of water bodies, and fluctuations in coastal currents.
Methods
Field
Plankton tows were conducted on 19 September 2012 in the southern channel, 1-2km offshore Heron Island. A global position system (GPS) was used to maintain consistent coordinates at 23’’27’538S, 151'' 556'47E. Samples were taken every 45 minutes during part of a tidal cycle, with two samples taken from before peak high on the flooding tide, one sample at peak high and three after peak high on the ebbing tide. The high tide peak was at 10.30am, and the low tide peak was 4.17am. At each sampling time point, three vertical tows were taken and consolidated into a labeled container, which was placed in a dark esky on ice. Each sample was taken from a depth of approximately 10metres, with a total depth of 20metres. The plankton net had a mesh size of 100µm, and a diameter of 30cm. A total of six samples were collected and returned to the laboratory for immediate microscopic analysis.
Laboratory
Each sample was gently swirled to obtain a homogenous mix and 5x10ml sub-samples were extracted using a 10ml pipette. Subsamples were placed into individual petrie dishes for analysis. Each subsample was examined under a dissecting microscope, and all creseidae found were extracted and placed in labeled specimen jars. The relative densities of C. acicula were the focus of this experiment, and counts of veligers and adult specimens of this species for each subsample were recorded. Individuals were observed and photographed and video recordings were taken of live animals using DinoXcope software.
Results
Qualitative
The planktonic community in the flooding samples (1 & 2) was comprised of primarily of larger organisms, including mysids, chaetognaths, larvaceans and copepods. Samples 3 (peak high tide) had large quantities of a thick mucosal substance, and particulate matter. Samples2 & 3 also had high abundances of Creseis recorded. Samples on the ebbing tide (4, 5, 6) had less trichodesmium, mucus and suspended particulate matter and much smaller planktonic organisms.
Quantitative
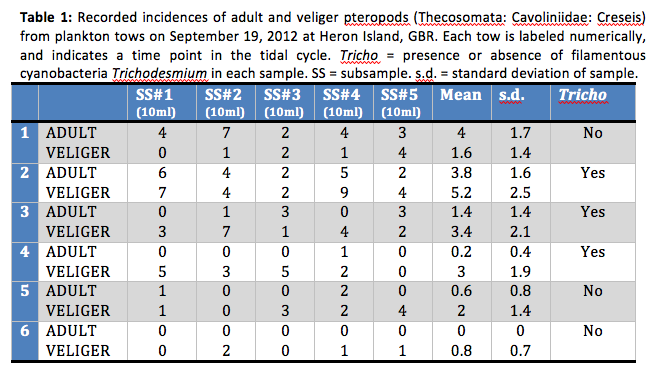
Discussion
The results of this study are inconclusive as very little can be inferred about planktonic distributions from one sampling trip. However some trends were observed that may instigate further study. The largest Creseis densities (adult and veliger) were found in association with thick densities of the filamentous cyanobacteria Trichodesmium. This was also the flooding high tide, with peak densities of both Creseis and Trichodesmium recorded in Sample 3 (peak high tide). Though not the initial objective of this study, it is an interesting phenomenon to note. Sakthivel &Haridas (1974) also observed associations between C. acicula ‘swarming’ and Trichodesmiumblooms in plankton tows taken on the west coast of India.
Trichodesmium is diazotrophic and plays a significant role in oligotrophic waters, as biological productivity is limited by the availability of nitrogen(N). Colonies of Trichodesmium have been found to support a unique microcosm, including bacteria, other cyanobacteria, protozoa, fungi, hydrozoans and copepods (Capone et al. 1997). C. acicula is known to feed on other phytoplanktonic organisms (diatoms, dinoflagellates, radiolarians and foramniferans), and not Trichodesmium directly. However, it is possible that the pteropods are secondarily exploiting these microcosms, capturing prey in their mucous webs. Further research is required to establish the extent of these Trichodesmium microcosms, and whether C. acicula may be feeding on them. This is particularly noteworthy, as the past decade has witnessed a significant increase in the frequency of cyanobacterial blooms along Queensland’s east coast over the past decade. This is possibly due to decreased water quality from anthropogenic pressures or may be an indicator of some other environmental change (Albert et al. 2005). Previous studies on the GBR have suggested that ‘new’ nitrogen introduced by cyanobacterial blooms could exceed those inputs from fluvial discharges, altering existing trophic dynamics (Bell et al. 2009).
This paper provides a comprehensive overview of the role of Trichodesmium and its significance to neritic zooplankton communities and global nitrogen cycles. |